oedema: excess interstitial fluid
- Formation – due to imbalance between hydrostatic and oncotic pressures not adequately balanced by lymph clearance
- Increased intravascular hydrostatic pressure – impaired venous return eg CCF, venous obstruction, constrictive pericarditis, ascites/cirrhosis, DVT
- Decreased extravascular hydrostatic pressure
- Decreased intravascular oncotic/osmotic pressure – nephrotic syn, malnutrition, protein losing enteropathy
- Increased extravascular oncotic/osmotic pressure – inflammation, sodium/water retention
- Decreased lymphatic return – neoplasia, infection, radiation, post surgery
Types
- Inflammatory – exudate(specific gravit >1.020)
- Cardiogenic – CCF –> dec CO–> dec renal perfusion –> renin/angiotensin sys–> salt/water retention–>increase blood volume and venous pressure. (transudate – specific gravity <1.012)
Haemostatic process: Sequence of events and Coagulation pathways.
- Injury leads to exposure of ECM and tissue factor
- Release of endothelin–> Transient arteriolar vasoconstriction (for haemostasis)
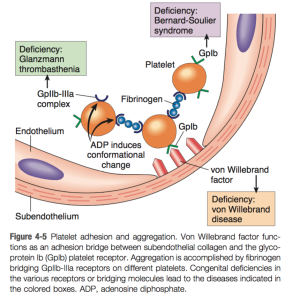
- Platelets adhere to ECM – GP1b receptors bind to vWF
- Platelets change shape as they become activated and they release granules(TXA2, ADP) – give sites clotting factors
- Platelets aggregate – ADP and TXA2 crucial
- Primary haemostatic plug
- The release of Ca from platelets speed up the coagulation cascade.
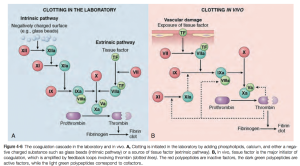
- Extrinsic pathway more common – tissue factor from injury site activates FVII –> Factor X–> Thrombin –> fibrinogen–>fibrin–> FXIII leads to crosslinking of fibrin
- Intrinsic pathway also activated –> finishes with fibrin
- Secondary haemostatic plug.
- Thrombin activates further platelet recruitment and granule release.
- Extrinsic Coagulation pathway
- Tissue factor/thromboplastin–>7 to 7a
- Intrinsic Coagulation Pathway
- XII –> XIIa
- XI–> Xia
- IX –> Ixa
- VIII–>VIIIa
- Common pathway
- X–>Xa
- II –> IIa (thrombin)
- Fibrinogen –>fibrin –>crosslinkined fibrin via XIIIa
Thrombosis:
Definition – inappropriate activation of normal haemostatic processes such as blood clot in a normal blood vessel
Pathogenesis – virchow’s triad any imbalance –> thrombus
- Hypercoagulability
- Endothelial injury
- Abnormal blood flow – turbulent, stasis eg AF
| Arterial | Venous | |
|---|---|---|
| Site |
|
Stasis |
| Growth | Retrograde | Direction of blood flow – tails can break off |
| Commonly occlusive | Invariably occlusive | |
| Common Sites | Coronary, cerebral, femoral | Lower limbs, sometimes in dural sinuses |
| Gray white, Lines of Zahn – alternating layers of platelets and fibrin and RBCs | Red/stasis thrombi with lots of RBCs |
Predisposing conditions for Thrombosis
- Primary/genetic
- Protein C and S deficiency
- Factor V leiden
- Prothrombin mutation
- Antithrombin deficiency
- Secondary/acquired
- SLE – antiphospholipid syndrome
- Prolonged bed rest
- Long distance travel
- Neoplasias
- DIC
- OCP
- Smoking
Potential outcomes of thrombosis
- Propagation –> Thrombotic occlusion
- Embolisation
- Resolution by dissolution
- Organisation and recanalisation – inflammation and fibrosis
DIC
Phenomenon of Excess release of prothrombotic agents into systemic circulation leading to widespread activation of thrombin and formation of microthrombi. This can lead to occlusion of end organs and associated ischaemia.
Concurrently the consumption of coagulation factors leads to coagulopathy and as a result a bleeding diathesis results.
Stimuli – Sepsis, Burns, Major trauma, Obstetric Cx such as FDIU, Amniotic fluid embolus
Embolism
Definition: detached intravascular solid, liquid or gaseous mass that is carried by blood distal from its sight of origin
Types & examples
- Thromboembolism – AF, DVT/PE
- Pulmonary
- dual blood supply so obstruction of medium sized arterioles –> just haemorrhag.
- Sudden death or cor pulmonale when 60% or more of circ is blocked
- Most are silent
- Repeat emboli –> pulm hypertension
- Systemic
- 2/3 are ventricular origin – AF or mural thrombi
- 75% lodge in lower limbs, 10% brain
- Pulmonary
- Air embolism
- Need in excess of 100ml to have clinical effect
- Obstetric procedures, chest wall injury, decompression sickness
- Amniotic fluid embolism
- Fat embolism – long bone fracture
- Pulmonary and cerebral occlusion
- Septic emboli
- FB
Infarction
- ischaemic necrosis of a tissue due to arterial or venous occlusion
- Types
- white infarction(usual solid organs) and
- red infarction(congested from double blood supply, venous occlusion),
- septic infarction
- Causes
- arterial occlusion from thrombus, embolism,
- hypoperfusion(systemic – cardiac failure, hypovolemia),
- vasoconstriction,
- extrinsic occlusion, torsion,
- traumatic rupture,
- entrapment in hernia,
- venous occlusion
- Factors the determine infarct development
- degree of collateral blood supply,
- oxygen demand/vulnerability of tissue,
- rate of occlusion,
- oxygen content of blood
Shock
- Definition – systemic hypoperfusion of tissues leading to cellular hypoxia
- Types – Cardiogenic · Hypovolaemic · Distributive (septic, anaphylaxis, spinal shock).
- Stages
- Non progressive – reflex compensation to perfuse major organs – baroreceptors, catecholamines, renin/angiotensin, ADH, sympathetic stimulation (Tachycardia, peripheral vasoconstriction and decreased urine production)
- Progressive – anaerobic glycolysis begins, lactic acidosis, and decreased vasomotor response –> vasodilation, peripheral pooling, hypoxic injury, prothrombotic, DIC, vital organ failure.
- Irreversible phase – lysosomal enzymes and NO released –> decreased myocardial contractility, ATN, bacteraemic shock from ischaemic gut.
- Lipopolysaccharide (LPS)
- at low dose –> local inflammation,
- at medium dose –> fever and systemic sx,
- at high dose –> prothrombotic, vasodilation and therefore shock.
- LPS starts a cytokine cascade of TNF, IL1, IL6/IL8
Anticlotting mechanisms
- After the secondary haemostatic plug is formed –> fibrinolytic pathways are initiated to balance the thrombus formation
- t-PA
- Urokinase like PA
- Plasminogen
- Thrombomodulin
- Anti thrombin III – inhibit thrombin and other intrinsic clotting factors
- Protein C and S – vit K dependent, block activated factor 5 and 8
- Tissue factor pathway inhibitor – stops the extrinsic coagulation pathway by inactivating Facto 7a and 10a.
- Limiting factor activation to sites of exposed phospholipids eg activated platelets.